Positive results using DehydraTECH-CBD support progressing to FDA IND application
KELOWNA, BC / September 8, 2021 / Lexaria Bioscience Corp. (NASDAQ:LEXX) (NASDAQ:LEXXW) (the "Company" or "Lexaria"), a global innovator in drug delivery platforms today announced it is formally beginning the process towards an Investigational New Drug ("IND") application filing with the Food and Drug Administration ("FDA") with its DehydraTECH-processed cannabidiol ("DehydraTECH-CBD") as a prospective registered pharmaceutical treatment for hypertension.
Lexaria has retained the services of an expert regulatory affairs and quality assurance consultancy group that will help prepare Lexaria for a pre-IND meeting with the FDA, as well as with designing the necessary non-clinical, clinical and related product development IND-enabling work to be completed in advance of the IND filing.
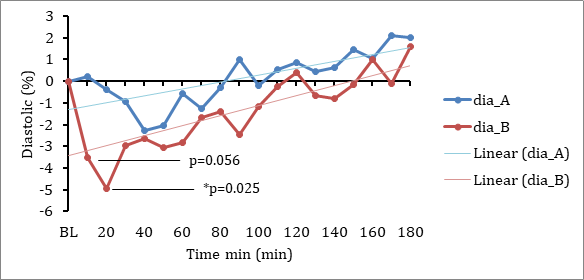
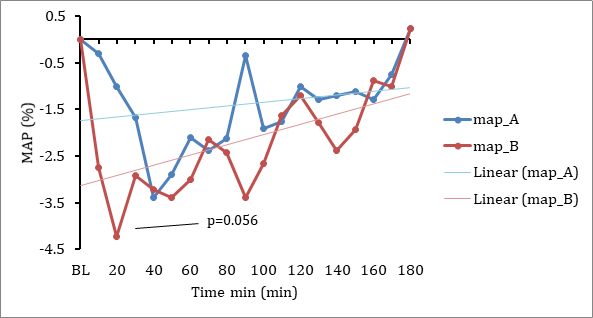
Lexaria's new IND enabling program is made possible through successfully completed studies that have provided support for more ambitious commercial goals. Recently achieved successful results from two 2021 human clinical pharmacodynamic hypertension studies and a 2018 human clinical pharmacokinetic ("PK") study, along with a number of successful animal studies demonstrating PK performance and molecular characterization work completed through Canada's National Research Council, have together established a strong body of evidence for Lexaria's DehydraTECH-CBD. These studies have shown that DehydraTECH-CBD demonstrates superior bioabsorption upon oral administration and is effective at reducing blood pressure with no significant unwanted side effects.
Provided that ongoing and upcoming studies continue to deliver favorable results, and that IND and other approvals from regulators are obtained, Lexaria intends to progress to a New Drug Application ("NDA") at the appropriate time, possibly via the abbreviated 505(b)(2) pathway.
The IND application process is also expected to utilize data from the Company's third and fourth 2021 human clinical hypertension studies, whereby Lexaria hopes that these studies will contribute further valuable data to its growing information package.
Lexaria will provide details on the timing and specifics of its planned pre-IND meeting and of the non-clinical, clinical and related product development IND-enabling work program elements to be completed in advance of and to culminate in IND filing as these details become available.
In addition, Lexaria is evaluating whether additional therapeutic indications for DehydraTECH-CBD might be pursued with the FDA in due course.
About DehydraTECH-CBD
DehydraTECH-CBD is a unique CBD formulation Lexaria has developed and is optimizing based on its patented and proprietary DehydraTECH drug delivery technology. DehydraTECH is designed to improve the way active molecules enter the bloodstream upon oral ingestion. DehydraTECH has also demonstrated enhanced delivery of certain active molecules including CBD into brain tissue, which Lexaria believes to be of particular importance for the effectiveness of its DehydraTECH-CBD specifically against hypertension because of the significant influence of central mediation upon blood pressure.
CBD in the Regulatory Environment.
CBD under the brand name Epidiolex® was approved by the FDA in June 2018 for treating certain types of pediatric seizure disorders. Recommended dosing of Epidiolex CBD is 2.5 mg per kilogram of bodyweight to begin treatment, taken twice per day, increasing to 5.0 mg per kilogram of bodyweight, taken twice per day. An average 9 year-old child weighs about 28 kg, so the FDA recommended dose of Epidiolex would initially be 140 mg, increasing to 280 mg daily for long term use. The average adult man in the US weighs 199.6 lbs (90.7kg), so if he was using CBD at that same rate, he would require 900 mg per day for a sustainable dose. The maximum FDA-approved dose of Epidiolex is 10.0 mg per kilogram of bodyweight, taken twice daily.
Hypertension Markets
The hypertension market is valued at $28 billion per year and is expected to continue growing as one of the world's top health problems. Geographically, some of the highest rates of growth are expected in more recently industrialized nations such as China and India. Over 1.1 billion people worldwide suffer from hypertension.
Fewer than 1 person in 4 with hypertension have successfully controlled their blood pressure through medications, meaning the potential market for hypertension drugs is much larger than $28 billion per year if an affordable drug was available with few or no side effects. Lexaria believes that its DehydraTECH-CBD may introduce a more tolerable anti-hypertensive treatment option that may be used alone or in combination with other medications, to reduce BP with fewer discouraging and unwanted side effects. Lexaria would seek to satisfy this currently unmet demand and in doing so could expand the overall hypertension market.
"Among persons 50 years of age or older, isolated systolic hypertension is the most common form of hypertension, and systolic blood pressure becomes more important than diastolic blood pressure as an independent risk predictor for coronary events, stroke, heart failure, and end-stage renal disease (ESRD). The Global Burden of Disease Study identified elevated blood pressure as the leading risk factor, among 67 studied, for death and disability-adjusted life-years lost during 2010."
Drugs focused on blood pressure and related conditions are some of the highest selling drugs in the world. Lipitor, used to treat high cholesterol and reduce the risk of heart disease, has generated $94.7 billion in revenue from 1992 until 2017. Plavix, used to prevent heart attack and stroke, has sold $46.5 billion from 1992 until 2017. There are several hypertension drugs that each generate $1 billion per year or more in revenue.
About Lexaria Bioscience Corp.
Lexaria Bioscience Corp.'s patented drug delivery technology, DehydraTECH™, improves the way active pharmaceutical ingredients (APIs) enter the bloodstream by promoting more effective oral delivery. Since 2016, DehydraTECH has repeatedly demonstrated the ability to increase bio-absorption with cannabinoids and nicotine by 5-10x and, in some instances with cannabinoids by as much as 27x compared to standard industry formulations, reduce time of onset from 1 - 2 hours to minutes, and mask unwanted tastes; and is also being evaluated for orally administered anti-viral drugs, non-steroidal anti-inflammatory drugs (NSAIDs), and more. DehydraTECH has also evidenced an ability to deliver some drugs more effectively across the blood brain barrier. Lexaria operates a licensed in-house research laboratory and holds a robust intellectual property portfolio with 21 patents granted in nations with a combined population of 2.6 billion, and over 50 additional patents pending worldwide. For more information, please visit www.lexariabioscience.com.
CAUTION REGARDING FORWARD-LOOKING STATEMENTS
This press release includes forward-looking statements as such term is defined under applicable securities laws. These statements may be identified by words such as "anticipate," "if," "believe," "plan," "estimate," "expect," "intend," "may," "could," "should," "will," and other similar expressions. Such forward-looking statements in this press release include, but are not limited to, statements by the company relating the Company's ability to carry out research initiatives, receive regulatory approvals or grants or experience positive effects or results from any research or study. Such forward-looking statements are estimates reflecting the Company's best judgment based upon current information and involve a number of risks and uncertainties, and there can be no assurance that the Company will actually achieve the plans, intentions, or expectations disclosed in these forward-looking statements. As such, you should not place undue reliance on these forward-looking statements. Factors which could cause actual results to differ materially from those estimated by the Company include, but are not limited to, government regulation and regulatory approvals or prohibitions, managing and maintaining growth, the effect of adverse publicity, litigation, competition, scientific discovery, the patent application and approval process, potential adverse effects arising from the testing or use of products utilizing the DehydraTECH technology, the Company's ability to maintain existing collaborations and realize the benefits thereof, delays or cancellations of planned R&D that could occur related to pandemics or for other reasons, and other factors which may be identified from time to time in the Company's public announcements and periodic filings with the US Securities and Exchange Commission on EDGAR. There is no assurance that any of Lexaria's postulated uses, benefits, or advantages for the patented and patent-pending technology will in fact be realized in any manner or in any part. No statement herein has been evaluated by the Food and Drug Administration (FDA). Lexaria-associated products are not intended to diagnose, treat, cure or prevent any disease. Any forward-looking statements contained in this release speak only as of the date hereof, and the Company expressly disclaims any obligation to update any forward-looking statements contained herein, whether as a result of any new information, future events, changed circumstances or otherwise, except as otherwise required by law.
INVESTOR CONTACT:
Phone: 250-765-6424, ext 202
SOURCE: Lexaria Bioscience Corp.
View source version on accesswire.com:
https://www.accesswire.com/662960/Lexaria-Begins-Investigational-New-Drug-IND-Enabling-Program-for-DehydraTECH-CBD-for-Hypertension